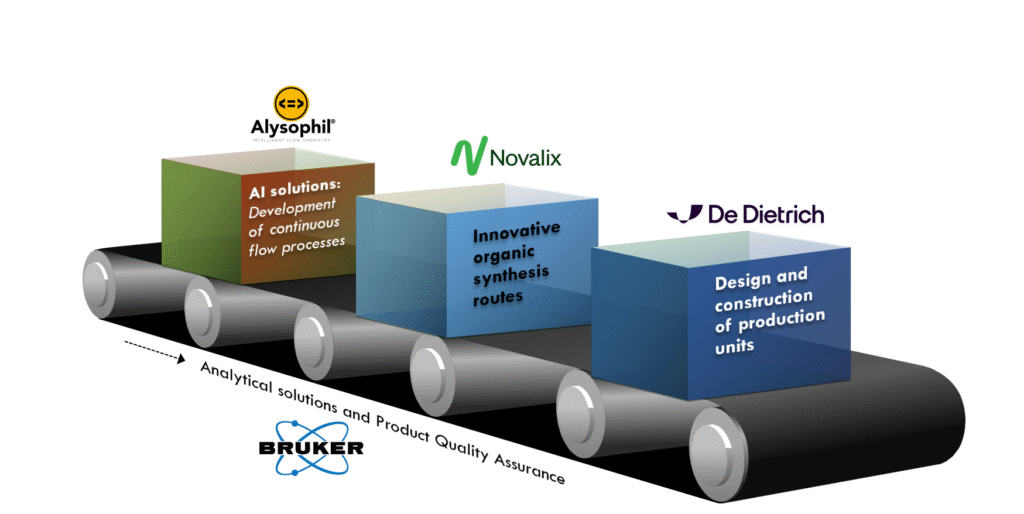
In the rapidly evolving world of pharmaceutical manufacturing, the integration of innovative technologies is essential for meeting the growing demand for active pharmaceutical ingredients (APIs). One such revolutionary approach is flow chemistry API production, which leverages continuous flow processes to enhance efficiency and scalability. As we navigate an era marked by unprecedented global health challenges, the need for robust and adaptable pharmaceutical production processes has never been more pressing. Recent developments have shown that embracing technologies like flow chemistry, artificial intelligence (AI), and nuclear magnetic resonance (NMR) can significantly streamline API production while ensuring high-quality standards. This article delves into the transformative potential of these technologies in API production, offering valuable insights into their benefits and applications.
Understanding Flow Chemistry API Production and its Advantages
Flow chemistry API production represents a shift from traditional batch processes, enabling continuous synthesis of pharmaceuticals. This approach offers several significant benefits:
- Enhanced Efficiency: Continuous processes minimize downtime and maximize throughput.
- Improved Safety: Reduced exposure to hazardous materials and better control of reaction conditions.
- Scalability: Flow chemistry allows seamless scaling from lab to industrial production without extensive re-engineering.
For instance, companies are increasingly adopting flow chemistry for developing complex molecules like fentanyl and propofol, where precise control over reaction parameters is crucial. This integrated approach also facilitates rapid process optimization, contributing to faster turnaround times for new drugs.
The Role of AI in Flow Chemistry API Production
Artificial intelligence is playing a transformative role in enhancing the capabilities of flow chemistry API production. By employing AI-driven technologies, pharmaceutical companies can achieve remarkable improvements:
- Real-Time Data Analytics: AI algorithms analyze production data in real-time, allowing for immediate adjustments to maintain optimal conditions.
- Predictive Maintenance: Integrating AI with predictive analytics helps anticipate equipment failures before they disrupt production.
For example, AI can be employed to monitor key performance indicators, ensuring that the production process remains within desired specifications. This proactive approach mitigates risks associated with batch variability and reduces waste.
Integrating NMR for Enhanced Quality Control
Nuclear magnetic resonance (NMR) technology is emerging as a critical component in the workflow of flow chemistry API production. The ability of in-line NMR to provide instantaneous feedback on product composition and quality enhances the robustness of manufacturing processes.
- Multi-Attribute Analysis: NMR can simultaneously measure multiple attributes, such as concentration, yield, and purity, ensuring comprehensive quality control.
- Streamlined Process Development: By integrating NMR with flow processes, researchers can rapidly optimize reaction conditions based on real-time data.
Combining NMR with AI can also enable a self-regulating production environment, significantly elevating overall product quality while reducing the need for manual oversight. The future of pharmaceutical manufacturing will increasingly rely on this synergy for achieving greater reliability and efficiency.
Challenges and Opportunities in Flow Chemistry API Production
Despite the advantages of flow chemistry API production, several challenges exist that manufacturers must address. Transitioning from traditional batch processes involves overcoming technical hurdles, regulatory considerations, and the integration of advanced technologies.
- Technical Limitations: Specific reactions may require further research to adapt them to continuous flow conditions.
- Regulatory Compliance: Meeting the stringent requirements of pharmaceutical regulations can complicate the implementation of new technologies.
However, these challenges present opportunities for innovation. Collaborations among pharmaceutical companies, technology providers, and regulatory bodies can help refine flow chemistry API production methods and establish best practices.
The Future of Flow Chemistry API Production
Looking ahead, the integration of flow chemistry API production, AI, and NMR will redefine the landscape of pharmaceutical manufacturing. As the demand for high-quality APIs continues to rise, industry players must remain agile and open to adopting new technologies.
- Smarter Manufacturing: The industry is moving toward more autonomous systems that leverage real-time analytics for optimization.
- Sustainability: Enhanced efficiency in production processes contributes to reduced waste and a smaller environmental footprint.
As these innovations mature, they promise to facilitate the production of essential medicines while adhering to the highest standards of quality.
To deepen this topic, check our detailed analyses on Nutrition & Diet section